Mission
Make dental device purchasing easier to inspect, easier to repeat, and easier to explain when a clinic, DSO, or distributor needs documentation before it can act.

Make dental device purchasing easier to inspect, easier to repeat, and easier to explain when a clinic, DSO, or distributor needs documentation before it can act.
By 2030, every Zimmer Biomet routed dental request will have a clean spec trail: product family, UDI reference, IFU path, and service owner in one packet.
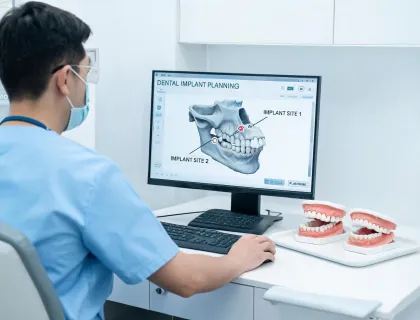



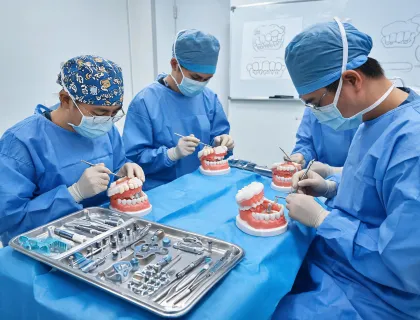
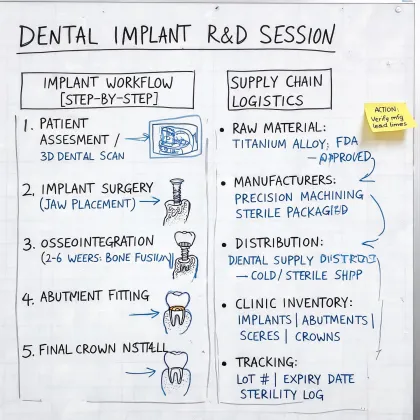
RA Specialist · Biomed FSE · Dental Clinical Application Specialist · Supply Documentation Lead · Sterile Packaging Coordinator
Talk to the Team